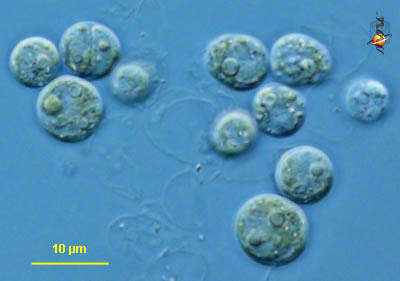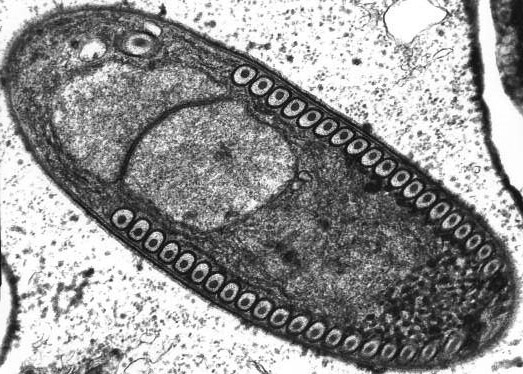
A microsporidian like this is one-half of the perp identified as the cause of Honeybee Colony Collapse Disorder. This isn't the exact species, but is instead another microsporidian called Fibrillanosema that infects amphipods. Neat rows of polar filament cross sections line either side like cannon on a British Man o' War. In 3D, these coil around in a big spiral. Creative Commons Leon White
For the last several years, honeybee colonies have been emptying out like keggers at which the cops have arrived — and the bees show every sign of being just as out of it as drunk college kids. They abandon their hives to die alone and cold in the wild, striking a huge blow to North American apiculture and, until now, anyway, leaving everyone scratching their heads.
Well, no longer. Though early reports identified an Israeli virus as one possible cause, a serendipitous bioinformatics project taken on by, of all people, the military, along with Montana researchers, has identified a dual cause of colony collapse: a previously unidentified DNA-virus, and a fungus called Nosema ceranae.
For those who’ve been paying attention, a Nosema parasite of bees has been around quite a long time — Nosema apis. But the new species, Nosema ceranae, appears to have come from Asia. Is this yet another introduced species decimation story? ( See Elm, American; Chestnut, American; and White Pine, Western) Or did we just never notice it here before? (the species can only be separated by DNA or scanning electron microscope — not by light microscope) Too early to tell. And still, no one knows what the fungus and virus combo is doing to make the bees lose it:
Still unsolved is what makes the bees fly off into the wild yonder at the point of death. One theory, Dr. Bromenshenk said, is that the viral-fungal combination disrupts memory or navigating skills and the bees simply get lost. Another possibility, he said, is a kind of insect insanity.
Translation: Zombification, a well known problem for insects under the influence of parasites (see Parasitoid wasps, and Cordyceps fungi)
Here’s the critical bit from the NYT article announcing the discovery:
Dr. Bromenshenk’s team at the University of Montana and Montana State University in Bozeman, working with the Army’s Edgewood Chemical Biological Center northeast of Baltimore, said in their jointly written paper that the virus-fungus one-two punch was found in every killed colony the group studied. Neither agent alone seems able to devastate; together, the research suggests, they are 100 percent fatal.
Since a 100 percent correlation seems pretty convincing, so let’s go with the assumption these guys are right for now. And since the virus is as yet unidentified and undescribed, let’s take a bit of a closer look at Nosema. Because Nosema is interesting. Really interesting. As recently as 10 years ago this group of organisms — colloquially known as microsporidia — was classified with next to Giardia, the most ancestral (aka primitive) nucleated (aka eukaryotic) organism known. Seriously. This would be like putting Homo sapiens at the base of the eukaryotic family tree*, because it turns out Nosema is a seriously evolved fungus. And in this case, as you’ll see, that crucial bit of taxonomic information makes a big practical difference in attacking this problem.
Microsporidians are all single-celled parasites that can infect nearly any eukaryotic host, but most have specialized on insects. They have a bunch of crazy standard features. First off, Nosema has no mitochondria, which is usually a requirement for self-respecting nucleated cells. Such nucleated cells, called the eukaryotes — or all life that isn’t bacteria or the similar looking archaea — employ tiny intra-cellular organelles called mitochondria to make energy using food and oxygen in the air. For the purposes of this post, we will ignore mitochondria’s fascinating origin story and incredible biochemical gymnastics and simply focus on the utter necessity of these organelles to the business of being alive for air-breathing eukaryotes (cyanide is fatal because it stops up the energy-producing works inside mitochondria), and how utterly strange it is that the otherwise unrelated groups of microsporidia, the parasite Giardia, and the amoebic parasite Entamoeba histolytica all lack them.
Instead, they possess an organelle called a mitosome, which seems to be a vestigial mitochondrion — that is, what’s left after the mitochondria are no longer necessary for cell upkeep and they start to degenerate to save the organism energy. Another example of vestigial and remnant structures might be the tiny leg bones still produced inside some snakes and whales — though these creatures’ ancestors stopped using their legs millennia ago, there is still a part a part of their genome that makes a much reduced and apparently energetically inconsequential vestige of them.
The reason parasites like these can get away with dispensing with their mitochondria is that precisely because they are parasites, they bathe in nutrients inside their host. They don’t need to breathe; their host does it for them. This seems to have happened several times in unrelated parasite groups. But because single-celled organisms possess few morphological characters, these groups were originally all placed together because they shared what few traits we could see: degenerate mitochondria (mitosomes), a double nucleus (microsporidia often have this, and Giardia always does — why is this adaptive for a parasite? No clue.), and a parasitic lifestyle. So a bunch of these funky parasites were thrown together into a classification called “archezoa“.
But someone must have studied these organisms’ DNA and found a very different story: microsporidia aren’t primitive protists, and aren’t related to Giardia and Entamoeba at all. They’re highly evolved fungi called zygomycetes — the same group that produces the bread mold Mucor and most snow molds that live at the foot of melting snowbanks that I wrote about in an August issue of High Country News. As recently as just 10 years ago, as printed in my copy of “The Variety of Life”, by Colin Tudge, they were still placed firmly at the base of the eukaryotic family tree — not near the tips of the branches, embedded in the fungi. It seems that the “archezoa” was really more of a niche than a true taxonomic grouping based on relatedness — these things evolved to occupy the same parasitic niches, and in the process, evolved similar adaptations, much as whales and fish look alike but come from very different sides of the tracks.
Zygomycetes are so called from the greek for “yoke” because they make a special sexual reproductive structure called a zygosporangium that yokes together two fungi of different mating types (=gender). As fungi, they follow the typical fungal body plan of being a bunch of thin filaments (aka mold). To look at a zygomycete, you would definitely think “fungus”. Not so with microsporidia! They tend to be single celled spores. But they also have cell walls made in part of chitin — another trait that unites the fungi.
But here’s the *really* weird thing about microsporidia. They are parasitic fungi that have evolved to look like protists and *act* like nematocytes (the stinging cells of jellyfish and anemones): Inside the spore is a coiled harpoon-like injection apparatus (go here to see it in 3D, rather than the 2D view at the top of this post) they use to get themselves into host cells. Just like nematocytes (covered in this post), the coiled “rope” of the harpoon turns inside out when the cell is triggered — and does so in less than 2 seconds. Once deployed, this long narrow filament (common size: .1-.2 micrometers in diameter by 50-500 micrometers long — click here to see one whose spring has sprung) inserts itself in a host cell and pumps the contents of the microsporidian inside. Pretty soon, the now zombified cell gets busy making little microsporidia.
Here’s the final important point, from the NYT:
They said that combination attacks in nature, like the virus and fungus involved in bee deaths, are quite common, and that one answer in protecting bee colonies might be to focus on the fungus — controllable with antifungal agents — especially when the virus is detected.
So without the taxonomic work to know these little jobbies are actually fungi and not protists, we wouldn’t know that we might have a chance of tackling the major threat to bees today with existing fungicides. Who says taxonomy is pointless?
____________________________________________________________________
* OK, maybe not quite. Since Nosema is a severely reduced parasite, it’s more like putting mistletoes — severely reduced parasitic plants — down there.